We are the agency who always gives you a priority on the free of question and you can easily make a question on the bunch.
FMTdb is a comprehensive and publicly accessible database that houses clinical information and sequencing data from donors and recipients who underwent Fecal Microbiota Transplantation (FMT) treatment for various functional gastrointestinal diseases. It serves as a valuable resource for researchers to explore and analyze the dynamic changes in gut microbiota, therapeutic efficacy, and treatment outcomes of FMT.
FMTdb includes three core types of standardized data:
(1) Clinical metadata: Demographics, medical history, laboratory results, screening records, treatment courses, and follow‑up outcomes for 15 donors and 370 recipients, plus 2,008 healthy controls.
(2) Microbiota sequencing data: 16S rRNA (V4 region) sequencing of fecal samples from donors (longitudinal multiple donations), recipients (pre‑FMT and post‑FMT), and healthy controls.
(3) Longitudinal profiles: Dynamic changes in donor microbiota across repeated donations and recipient microbiota across multiple FMT courses.
FMTdb focuses on functional gastrointestinal disorders (FGIDs) and related refractory gastrointestinal diseases, including:
Functional gastrointestinal disorders: chronic functional constipation, functional diarrhea, irritable bowel syndrome (IBS)
Infectious diarrhea: recurrent Clostridioides difficile infection (rCDI)
Inflammatory bowel disease (IBD): Crohn’s disease (CD), ulcerative colitis (UC)
Other conditions: radiation enteritis, incomplete intestinal obstruction, and FGIDs with comorbidities (e.g., anxiety).
Efficacy is assessed by a team of experienced physicians based on gastrointestinal symptom improvement.
• Response: clinical relief or cure of symptoms.
• Non‑response: no significant improvement.
Follow‑ups are performed at 1, 3, 6, and 12 months post‑treatment. Efficacy is stratified by disease type and donor to reveal donor‑dependent and disease‑dependent response patterns.
FMTdb provides interactive visualization and analysis tools:
1. Donor–recipient matching network: Visualize donor–recipient pairs and efficacy distribution.
2. Longitudinal microbiota tracking: Dynamics of donor microbiota across repeated donations.
3. Recipient microbiota dynamics: Pre‑FMT vs. post‑FMT changes and donor strain engraftment (“donorization”).
4. Microbiota comparison: Between patients and healthy controls, and between high‑/low‑efficacy donors.
5. Immunological correlation: Changes in lymphocyte subsets and inflammatory markers pre‑ and post‑FMT.
FMTdb is publicly available via the official web portal: https://gift2disease.net/FMTdb/
All users must agree to the FMTdb Data Usage Agreement (DUA). All participant data are de‑identified; re‑identification and redistribution are strictly prohibited.
Code availability: Analyses used QIIME2 (v2023.2.0), DADA2, and R (v4.3.1). Custom pipelines: https://github.com/FMTdb/FMTdb_pipeline
Raw sequencing data: deposited in NCBI SRA (PRJNA1061457, PRJNA1061329) and CNCB GSA (PRJCA031558).
Processed data: De‑identified clinical and microbial metadata are available via the FMTdb portal under controlled access.
The interface is intuitive and organized into core modules:
• Donor Information: donor screening, longitudinal microbiota, and recipient response rates
• Recipient Information: diagnosis, treatment history, pre‑/post‑FMT microbiota, and outcomes
• Flora Dynamics: time‑series microbiota abundance changes
• Data Download: ethics‑approved processed datasets
• Help & Support: user guidance and contact
All tools support one‑click interactive analysis:
• View donor–recipient matching networks and efficacy stratification.
• Track longitudinal microbiota stability in donors.
• Compare pre‑FMT vs. post‑FMT microbiota and engraftment patterns.
• Analyze differences between patients and healthy controls.
Tooltips and on‑page instructions guide each step.
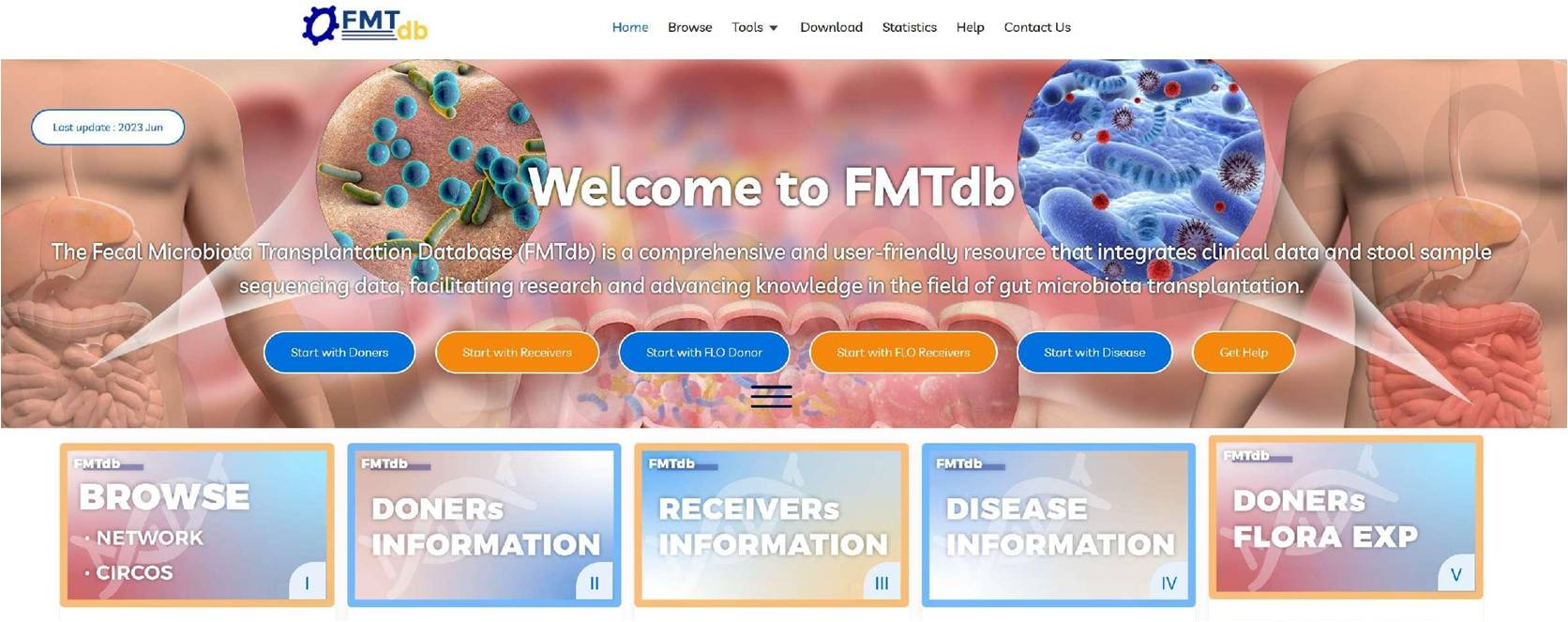
The homepage provides quick access to donor‑centered, recipient‑centered, and microbiota‑centered visualization and analysis.
Users can download processed, ethics‑approved data for further independent analysis.
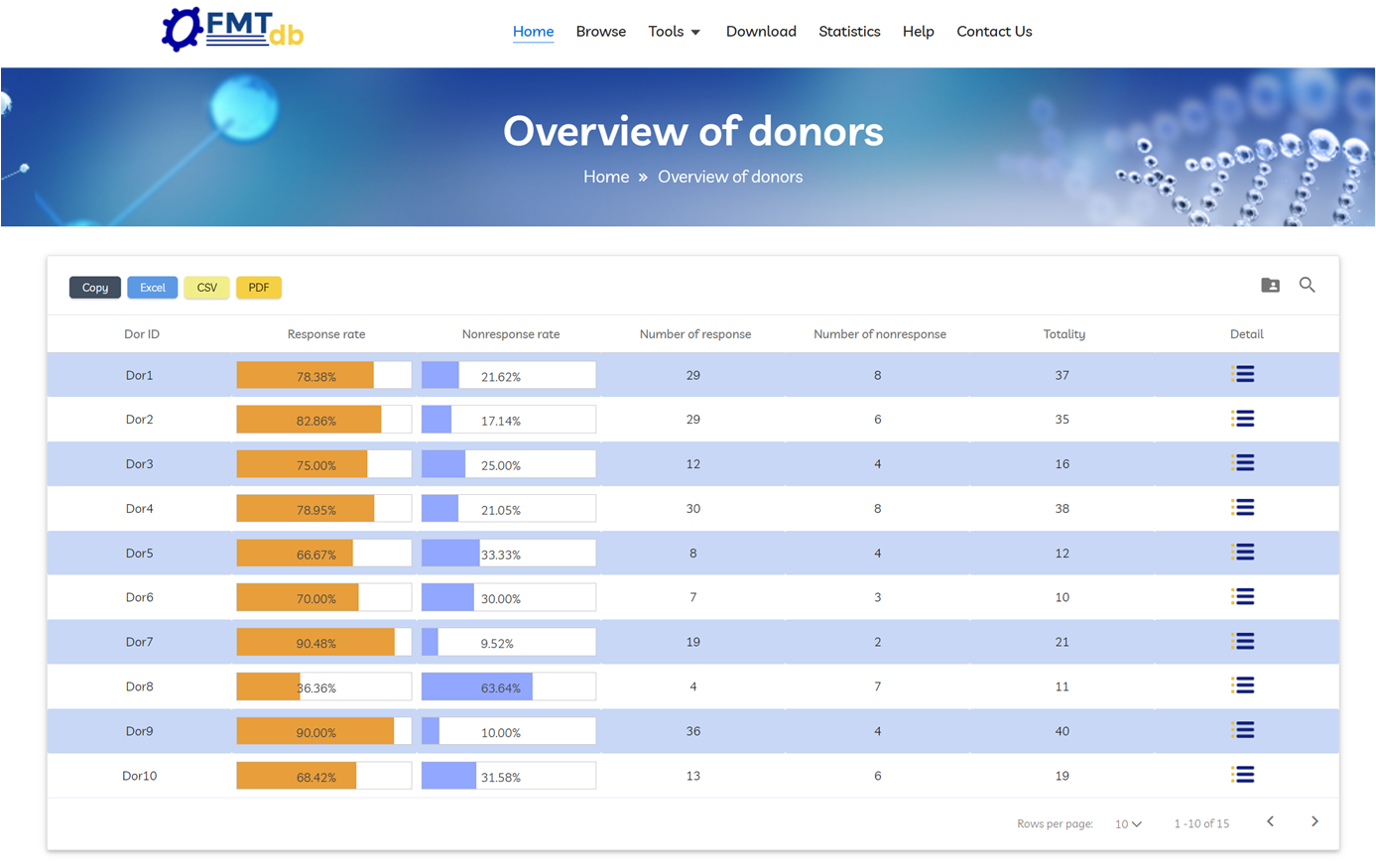
Click “Start with Donors” to view the overall response rate and non‑response rate for each donor. Click “Detail” to view full donor profiles.
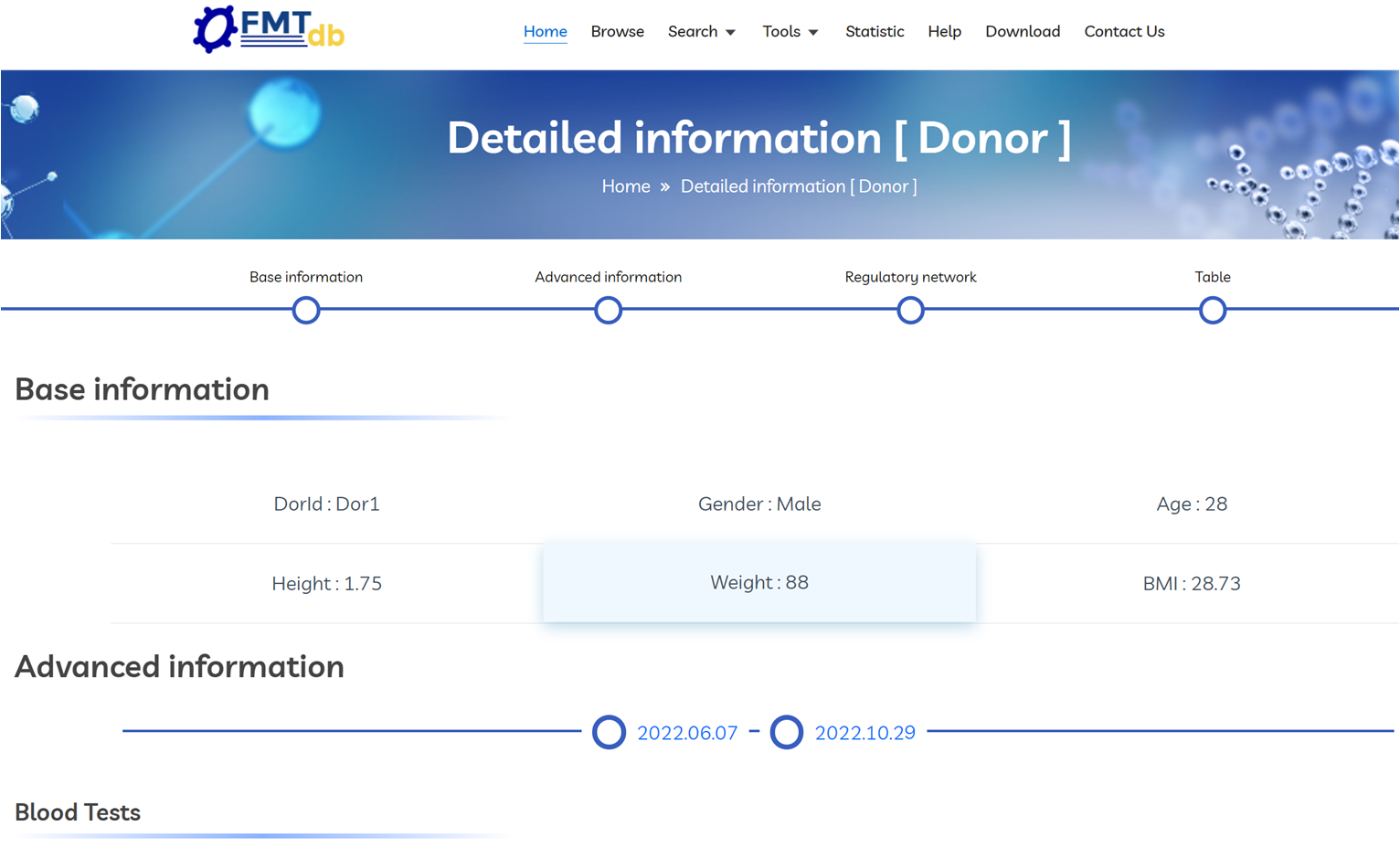
The donor detail page includes: basic demographics, screening and laboratory results, recipient and disease distribution, longitudinal microbial composition across donations, and treatment efficacy summary.
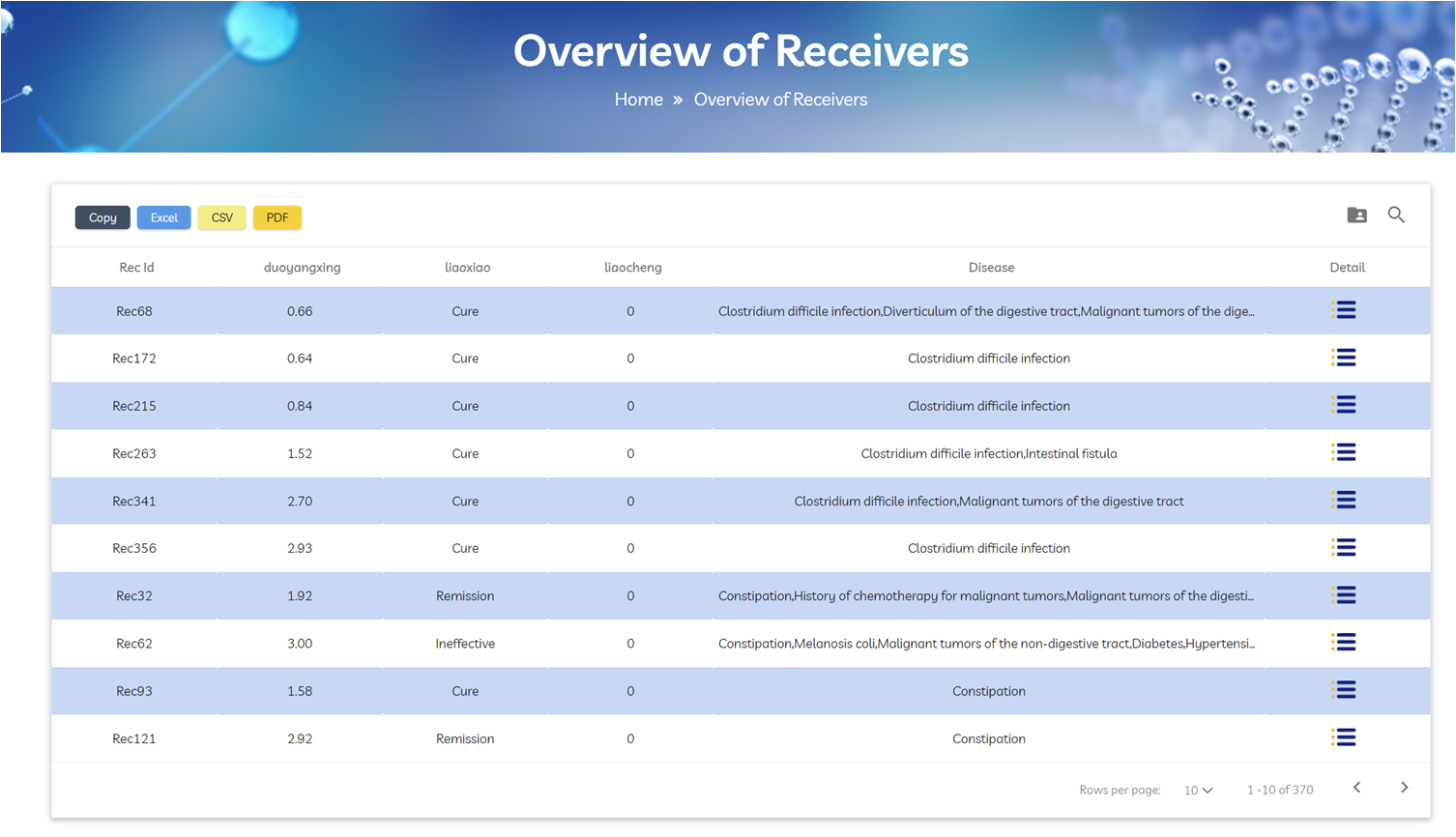
Click “Start with Receivers” to browse diagnosis, treatment courses, and efficacy status for each recipient. Click “Detail” to view pre‑FMT baseline, post‑FMT changes, microbiota dynamics, and follow‑up data.
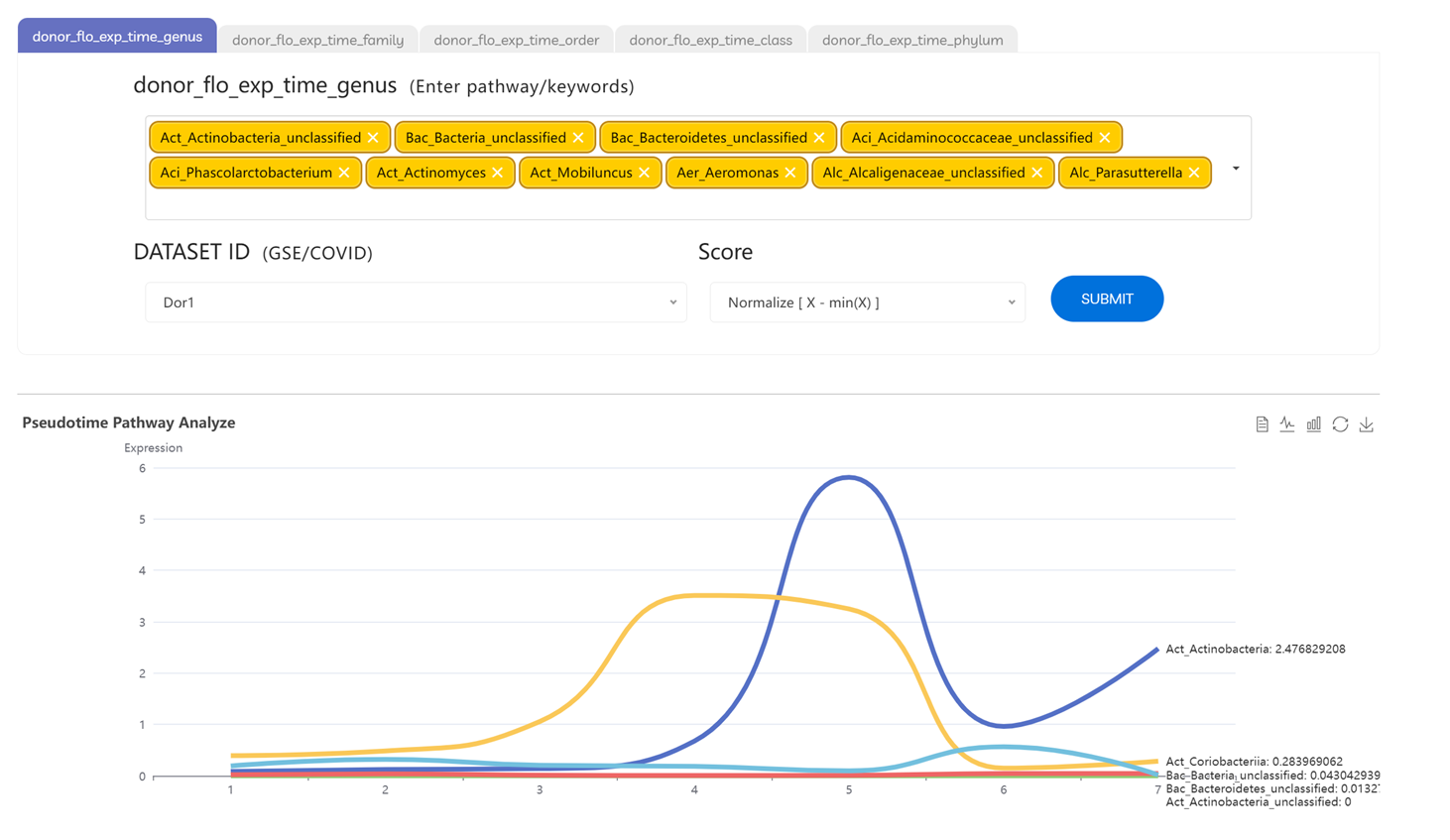
Click “Start with FLO” to view longitudinal changes in gut microbiota abundance for donors or recipients across multiple time points or treatment courses, showing stability, fluctuations, and engraftment trends.
The study was approved by the Ethics Committee of Shanghai Tenth People’s Hospital. All participants provided written informed consent. All personal identifiers are anonymized and securely stored. Users must comply with data usage policies and ethical requirements.
We welcome user feedback, questions, and collaboration inquiries. Please contact the development team via the contact information on the FMTdb website. Your input helps us improve data quality, functions, and user experience.